March 23, 2013 8:56 pm
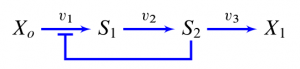
Choke points, load points, hot spots, key steps and critical steps are some of the many phrases that have been coined to describe places in a biochemical network where perturbations are said to make a difference. Locating these places is important because they are sites where we would target a drug or reengineer to change a cell's phenotype. The question is what are these points, is a choke point something that can be identified as a physical thing, can a choke point be removed from a pathway and studied in isolation? To answer these questions let us look at a very simple two step pathway. Both steps in the pathway are governed by reversible Michaelis-Menten kinetics, so nothing out of the ordinary. Let's say that a perturbation to the enzyme activity at the second step results in a significant change in the steady-state flux through our little pathway, The first step has hardly any effect. We might therefore call the second step the choke point of the pathway, or the rate-limiting step, the hot spot etc.
Is there something special about the second step that makes it a 'choke point'? What may surprise many is that it is the first step that makes the second step a choke point. There is nothing intrinsically special about the second step that makes it the choke point, all the interesting action occurs at the first step.
The explanation is simple. The reaction rate through the first step is very sensitive to the product it makes. If the product of the first step goes up a bit the result is that the reaction rate through the first step goes down, this is simply due to strong product inhibition, that is the product is an effective competitor with respect to the substrate for the first step. So what? Consider this scenario, we add a drug that can inhibit the activity of the second step. Its reaction rate goes down. This results in the substrate for the second step to go up (its being consumed less so it must go up). Since the first step is sensitive to this concentration, its rate goes down. The net effect is that it looks as if the second step is the choke point. The name choke suggests a small diameter pipe so that making the pipe smaller has a marked effect on the flow. But this isn't what is happening here. What is happening is that there is negative feedback from the second step to the first step and it is the first step that slows down the flow. If it weren't for the product inhibition on the second step, the second step would have NO effect on the flow. The second step is actually NOT a choke point at all.
The words we use can be very misleading especially when dealing with complex systems. In this case the work choke is extremely misleading. What about the other phrases that people use, such as a hot spot. Hot spot is certainly a colorful phrase to use but is it a phrase that should be used by intelligent people? Its certainly a word that politicians or my 4 your old might understand, but is it a word that professional scientists should use? What about the word key step or critical step? These don't convey the point that the step itself is not the one responsible for it being influential, it was the first step that gave the second step its ability to control flux. If anything the key step is the first step. These words aren't therefore really appropriate either. What about the word rate-limiting step, still in use today in some circles? That word also suggests a pipe that is too small to carry the flow, it has the same problem as the word choke point.
Most of the words we use are either imprecise or just misleading. What do we use instead? As scientists we should define precisely what we mean, preferably operationally and quantitatively. Forty years ago such a definition was given, that definition is the flux control coefficient, it is simply a number that tells us how much of an effect a perturbation has on the system as a whole. It doesn't explain why a particular step has a given a degree of influence, it just tells us how much influence the step has.
Metabolic pathways such as the amino acid biosynthetic pathways which have allosteric negative feedback are an interesting case. The old literature (and even some new literature) will refer to the step that is regulated by the allosteric effector the rate-limiting step, the choke point or hot spot in more recent language. But modifying this step actually has little effect on the phenotype. It turns out that its the step after the feedback loop that has most of the influence. However it is the regulated step that allows influence to be on the last step. The allosterically regulated step is an important step but it certainly isn't the rate-limiting step, choke point or hot spot.
A real choke point, The Battle of Thermopylae.
Leonidas at Thermopylae, by Jacques-Louis David, 1814.
Image and data provided by Columbia University.